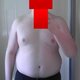
how much more before i can get laid?
edit: **** you all
|
|
Thread: Do i even lift? Picture
-
04-15-2013, 02:14 PM #1
-
04-15-2013, 02:16 PM #2
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
04-15-2013, 02:17 PM #3not even my final form

- Join Date: Jul 2007
- Location: California, United States
- Posts: 1,598
- Rep Power: 3235












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Human Spaceflight Engineer
"The sharp edge of a razor is difficult to pass over; thus the wise say the path to Salvation is hard"
-
04-15-2013, 02:18 PM #4
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.***Georgia crew***
-
-
04-15-2013, 02:19 PM #5
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.++ Positive Crew ++
"As you think, so shall you become."
-
04-15-2013, 02:19 PM #6
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
04-15-2013, 02:19 PM #7
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Protect Ya Neck
-
04-15-2013, 02:20 PM #8
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
-
04-15-2013, 02:21 PM #9Kava Head

- Join Date: Nov 2012
- Location: Galveston, Texas, United States
- Age: 39
- Posts: 2,316
- Rep Power: 1772












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Lift, Run, Surf, Kava.
For Your Health.
Current Lifts:
Bench: 320x1
Squat: 495x1
Dead: 545x1
*never uses hover crew*
-
04-15-2013, 02:21 PM #10
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Angels
-
04-15-2013, 02:21 PM #11
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
04-15-2013, 02:22 PM #12
Well, Quantum mechanics (QM � also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave�particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
-
04-15-2013, 02:22 PM #13
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
04-15-2013, 02:22 PM #14Registered User

- Join Date: Sep 2012
- Location: Brookville, Indiana, United States
- Age: 28
- Posts: 1,516
- Rep Power: 1392












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects*always pick 7 crew*
*WetBreasts is gonna make it crew*
*Self-Improvement Crew*
-
04-15-2013, 02:22 PM #15
-
04-15-2013, 02:23 PM #16The World Is Yours

- Join Date: Nov 2012
- Location: United Kingdom (Great Britain)
- Posts: 2,357
- Rep Power: 1481












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
Edit: in on first page of epic thread. I think it's time you get a serious answer OP, here goes.....
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Last edited by Samevans7; 04-16-2013 at 01:46 AM.

-
-
04-15-2013, 02:23 PM #17
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects
-
04-15-2013, 02:23 PM #18
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects[300 Spartan Helot]
Scared of Clowns and Dolls crew
Works at Chipotle crew
Eat Chipotle errr day crew
Twin brother crew (PJZBoozer)
Chicano Brah crew
-
04-15-2013, 02:24 PM #19
sʇɔǝɾqo ǝlɐɔs-ɔıɯoʇɐ ɹǝɥʇo puɐ 'suoɹʇɔǝlǝ 'suoʇoɥd ɟo ɹoıʌɐɥǝq ǝɥʇ ɟo ʍǝıʌ pǝıɟıun ɐ ǝpıʌoɹd ǝldıɔuıɹd ʎʇuıɐʇɹǝɔun ǝɥʇ puɐ ɹǝʇʇɐɯ puɐ ʎƃɹǝuǝ ɟo ʎʇılɐnp ǝlɔıʇɹɐd–ǝʌɐʍ ǝɥʇ 'sɔıuɐɥɔǝɯ ɯnʇuɐnb ɟo ʇxǝʇuoɔ ǝɥʇ uı ˙pǝzıʇuɐnb sı ǝlnɔǝloɯ ɹo ɯoʇɐ uɐ oʇ punoq uoɹʇɔǝlǝ uɐ ɟo ɯnʇuǝɯoɯ ɹɐlnƃuɐ ǝɥʇ 'ǝldɯɐxǝ ɹoɟ ˙ʎɐʍ (ƃolɐuɐ ˙ɟɔ) snonuıʇuoɔ ɐ uı ʇou puɐ '(ɐʇuɐnb uıʇɐl) sʇunoɯɐ ǝʇǝɹɔsıp uı ʎluo ǝƃuɐɥɔ uɐɔ sǝıʇıʇuɐnb lɐɔısʎɥd ǝɯos ʇɐɥʇ uoıʇɐʌɹǝsqo ǝɥʇ ɯoɹɟ sǝʌıɹǝp sɔıuɐɥɔǝɯ ɯnʇuɐnb ǝɯɐu ǝɥʇ [pǝpǝǝu uoıʇɐʇıɔ]˙sǝɹnʇɐɹǝdɯǝʇ ɹo sǝıƃɹǝuǝ (ɥƃıɥ ʎɹǝʌ ɹo ʍol ʎɹǝʌ '˙ǝ˙ı) ǝɯǝɹʇxǝ ʎluo ʇɐ ǝƃɹǝɯǝ puɐ ɔıdoɔsoɹɔɐɯ ǝɹɐ sɹoıʌɐɥǝq ǝsǝɥʇ ɟo ǝɯos 'sɔıuɐɥɔǝɯ ɯnʇuɐnb ɟo sɔıdoʇ pǝɔuɐʌpɐ uı
˙ɹǝʇʇɐɯ puɐ ʎƃɹǝuǝ ɟo suoıʇɔɐɹǝʇuı puɐ ɹoıʌɐɥǝq ǝʞıl-ǝʌɐʍ puɐ ǝʞıl-ǝlɔıʇɹɐd lɐnp ǝɥʇ ɟo ɥɔnɯ ɟo uoıʇdıɹɔsǝp lɐɔıʇɐɯǝɥʇɐɯ ɐ sǝpıʌoɹd sɔıuɐɥɔǝɯ ɯnʇuɐnb ˙sǝlɐɔs ɥʇƃuǝl ɔıɯoʇɐqns puɐ ɔıɯoʇɐ ɟo ɯlɐǝɹ ɯnʇuɐnb ǝɥʇ ʇɐ ʎlıɹɐɯıɹd sɔıuɐɥɔǝɯ lɐɔıssɐlɔ ɯoɹɟ sʇɹɐdǝp sɔıuɐɥɔǝɯ ɯnʇuɐnb ˙ʇuɐʇsuoɔ ʞɔuɐld ǝɥʇ ɟo ɹǝpɹo ǝɥʇ uo sı uoıʇɔɐ ǝɥʇ ǝɹǝɥʍ 'sǝlɐɔs ɔıdoɔsoɹɔıɯ ʇɐ ɐuǝɯouǝɥd lɐɔısʎɥd ɥʇıʍ slɐǝp ɥɔıɥʍ sɔısʎɥd ɟo ɥɔuɐɹq ɐ sı (ʎɹoǝɥʇ ɯnʇuɐnb ɹo 'sɔısʎɥd ɯnʇuɐnb sɐ uʍouʞ oslɐ – ɯb) sɔıuɐɥɔǝɯ ɯnʇuɐnb 'llǝʍgloots for the sloots
*so alpha cant hold eye contact with self in mirror crew*
-
04-15-2013, 02:24 PM #20
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
-
04-15-2013, 02:24 PM #21
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objectsWill Put in Reps of hard work for Rep
Laurier Uni Brah lulz
*MISC TORONTO CREW*
-
04-15-2013, 02:24 PM #22Have I ever even lifted?

- Join Date: Oct 2010
- Location: Calgary, Alberta, Canada
- Posts: 4,908
- Rep Power: 624












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.*WetBreast is gonna make it crew*
-
04-15-2013, 02:25 PM #23
Well, Quantum mechanics (QM � also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave�particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
Well, Quantum mechanics (QM � also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave�particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.Short-term goals
Make the decision to start loosing weight (15th April'13) [X]
=<250lbs by may 1st [10 lbs lost] []
=<240lbs by June 1st [20 lbs lost] []
=<230lbs by july 1st [30 lbs lost] []
=<220lbs by aug 1st [40 lbs lost] []
=<210lbs by sep 1st [50 lbs lost] []
=<200lbs by oct 1st [60 lbs lost] []
Long-term Goal
185lbs @ 8-10BF% [75 lbs lost] []
-
04-15-2013, 02:25 PM #24Go Gata

- Join Date: Jul 2010
- Location: Miami, Florida, United States
- Age: 31
- Posts: 4,419
- Rep Power: 1883












Pues bien, la mec�nica cu�ntica (QM - tambi�n conocida como la f�sica cu�ntica o la teor�a cu�ntica) es una rama de la f�sica que se ocupa de los fen�menos f�sicos a escalas microsc�picas, donde la acci�n es del orden de la constante de Planck. La mec�nica cu�ntica se aparta de la mec�nica cl�sica, ante todo en el dominio cu�ntico de escalas de longitud at�mica y subat�mica. La mec�nica cu�ntica proporciona una descripci�n matem�tica de gran parte del comportamiento dual de part�culas y como ondas perfectas y las interacciones de la energ�a y la materia.
En temas avanzados de la mec�nica cu�ntica, algunos de estos comportamientos son macrosc�picos y s�lo emergen a extremos (es decir, muy bajo o muy alto) energ�as o temperaturas. [Cita requerida] El nombre de la mec�nica cu�ntica se deriva de la observaci�n de que algunas cantidades f�sicas pueden cambiar s�lo en cantidades discretas (cuantos de Am�rica), y no en un proceso continuo (cf. anal�gica) forma. Por ejemplo, el momento angular de un electr�n ligado a un �tomo o mol�cula est� cuantizada. En el contexto de la mec�nica cu�ntica, la dualidad onda-part�cula de la energ�a y la materia y el principio de incertidumbre de proporcionar una visi�n unificada del comportamiento de los fotones, electrones, y otros objetos a escala at�mica☆☆☆FLORIDA GATORS CREW☆☆☆
+++HEAT NATION+++
ΠΚΑ
�To create more positive results in your life, replace �if only� with �next time�.�
-
-
04-15-2013, 02:26 PM #25
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way.. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
04-15-2013, 02:26 PM #26
Many often confuse the difference between sex and making love. Some often compare the two as being the same thing. Most people do not realize that there is a difference between these two. In this article I will give my opinion on why these two things are different from one another.
Let's start with sex. To me sex is something that doesn't often involve real feelings or even a committed, real relationship. Many people have a "friend with benefits," which is what I would put under the category of sex. Often times there are others who seek pleasure from one-night stands. There is never any real emotions or deep feelings; it's ONLY about the pleasure of the act. Usually when people only seek to have sex, there is very little fore play and intimacy afterwards, the holding and cuddling of one another, well that don't happen often. Many people are fine with having sex just for pleasure, never caring for the emotional aspects of being in love. Often some will jump partner to partner because that is all they are seeking, which is the immediate rush and release in the pleasure of sex.
-
04-15-2013, 02:26 PM #27
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects
Pues bien, la mec�nica cu�ntica (QM - tambi�n conocida como la f�sica cu�ntica o la teor�a cu�ntica) es una rama de la f�sica que se ocupa de los fen�menos f�sicos a escalas microsc�picas, donde la acci�n es del orden de la constante de Planck. La mec�nica cu�ntica se aparta de la mec�nica cl�sica, ante todo en el dominio cu�ntico de escalas de longitud at�mica y subat�mica. La mec�nica cu�ntica proporciona una descripci�n matem�tica de gran parte del comportamiento dual de part�culas y como ondas perfectas y las interacciones de la energ�a y la materia.
-
04-15-2013, 02:26 PM #28
Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way.. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.
-
-
04-15-2013, 02:27 PM #29
Nun, Quantenmechanik (QM - auch wie die Quantenphysik oder der Quantentheorie bekannt) ist ein Teilgebiet der Physik, die mit physikalischen Ph�nomenen auf mikroskopischer Skala, wo die Action ist in der Gr��enordnung der Planck-Konstante besch�ftigt. Quantenmechanik weicht von der klassischen Mechanik in erster Linie an der Quanten-Bereich der atomaren und subatomaren L�ngenskalen. Die Quantenmechanik liefert eine mathematische Beschreibung der Gro�teil der dualen teilchenartige und Wave-�hnliches Verhalten und Interaktionen von Energie und Materie.
In fortgeschrittenen Themen der Quantenmechanik, sind einige dieser Verhaltensweisen makroskopischen und ergeben nur extreme (dh sehr niedrige oder sehr hohe) Energien oder Temperaturen. [Bearbeiten] Der Name der Quantenmechanik ergibt sich aus der Beobachtung, dass einige physikalische Gr��en k�nnen nur in diskreten Mengen (Latin Quanten), und nicht in einem kontinuierlichen (vgl. analog) Weg. Zum Beispiel wird der Drehimpuls eines Elektrons gebunden an ein Atom oder Molek�l quantisiert. Im Rahmen der Quantenmechanik, stellen die Welle-Teilchen-Dualismus von Materie und Energie und die Unsch�rferelation eine einheitliche Sicht auf das Verhalten von Photonen, Elektronen und anderen atomaren Objekten.Angels
-
04-15-2013, 02:28 PM #30Registered User

- Join Date: May 2009
- Location: Calgary, Alberta, Canada
- Age: 30
- Posts: 909
- Rep Power: 2903












Well, Quantum mechanics (QM – also known as quantum physics, or quantum theory) is a branch of physics which deals with physical phenomena at microscopic scales, where the action is on the order of the Planck constant. Quantum mechanics departs from classical mechanics primarily at the quantum realm of atomic and subatomic length scales. Quantum mechanics provides a mathematical description of much of the dual particle-like and wave-like behavior and interactions of energy and matter.
In advanced topics of quantum mechanics, some of these behaviors are macroscopic and emerge at only extreme (i.e., very low or very high) energies or temperatures.[citation needed] The name quantum mechanics derives from the observation that some physical quantities can change only in discrete amounts (Latin quanta), and not in a continuous (cf. analog) way. For example, the angular momentum of an electron bound to an atom or molecule is quantized. In the context of quantum mechanics, the wave–particle duality of energy and matter and the uncertainty principle provide a unified view of the behavior of photons, electrons, and other atomic-scale objects.***Canadian Crew***























Bookmarks