This info is for the newbies, again most veterans members have this knowledge. If you have any info on this matter please add it. Thanks
http://www.innvista.com/health/nutri...s/electrol.htmElectrolytes
ELECTROLYTES are chemical substances that separate, when dissolved in fluids, into electrically charged particles (ions) capable of conducting electric currents vital for the function of nerves and muscles.
The major cations (positively charged electroylytes) in the body's fluid supply are sodium, potassium, calcium, and magnesium. The major anions (negatively charged electrolytes) are chloride, phosphate, sulfate, and bicarbonate. Organic acids such as lactate, pyruvate, and aceto-acetate also carry negative ions.
Proteins are polyelectrolytes in that they carry many charges and may either be positively or negatively charged. In extracellular fluid, sodium is the major electrolyte with potassium present in much lower concentrations. The concentration of sodium can be 15 times higher than potassium in the extracellular fluid. Within the intracellular fluid, the situation is reversed with the major electrolyte being potassium while sodium is found in lesser concentrations. Here, concentrations of potassium can be 30 times greater than sodium.
Stability of electrolyte balance is dependent upon several factors. One is an adequate intake of water. Electrolytes are involved in metabolic activities and are vital to the function of all cells, therefore, when the body becomes dehydrated, electrolytes do not have sufficient fluid for mobility in order to function. Another factor is an adequate intake of electrolyte-containing foods. Still another is the homeostatic conditions within the body that can regulate the absorption, distribution, and excretion of water and its dissolved particles. Imbalances can occur when serum levels become either too high or too low usually resulting from renal disease or prolonged vomiting or diarrhea.
The following are major electrolytes and their functions:
Sodium creates much of the osmotic pressure of extracellular fluid and is the most abundant cation in it. It is essential for electrical activity of neurons and muscle cells. An imbalance can cause hyponatremia or hypernatremia. Hyponatremia results when blood levels of sodium become too low usually caused by excessive sweating, diarrhea, or vomiting. Symptoms are dizziness, confusion, weakness, low blood pressure, and shock. Hypernatremia happens when the sodium blood levels become too high as a result of an excessive water loss or sodium ingestion. Symptoms are extreme thirst and agitation.
Potassium creates much of the osmotic pressure in intracellular fluid and is the most abundant cation in it. It is essential for electrical activity of neurons and muscle cells. An imbalance can produce hypokalemia or hyperkalemia. Hypokalemia results when blood levels of potassium become too low and is usually a consequence of vomiting, diarrhea, or kidney disease. Symptoms are fatigue, confusion, and possible cardiac arrest. Hyperkalemia comes as a result of blood levels of potassium becoming too high and is usually a consequence of Addison's Disease with symptoms of weakness, abnormal sensations, cardiac arrhythmias, and possible arrest.
Calcium is found most abundantly (98%) in bones and teeth with the remaining in tissues and fluids. It maintains normal excitability of neuons and muscle cells and is essential for blood clotting. An imbalance causes hypocalcemia or hypercalcemia. Hypocalcemia occurs when the blood levels of calcium become too low usually as a result of a decreased function of the parathyroid gland or a decreased calcium intake. Symptoms include muscle spasms leading to tetany (a continuous spasm). Hypercalcemia occurs when the parathyroid over functions. Such symptoms include muscle weakness, bone fragility, and possibly kidney stones.
Magnesium is concentrated mostly in bone. It functions mainly in intracellular fluid rather than extracellular. It is essential for ATP production and activity of neurons and muscle cells.
Chloride is the most abundant anion in extracellular fluid. It easily diffuses into and out of cells helping to regulate osmotic pressure. It is also part of the hydrochloric acid in the stomach.
Bicarbonate is part of the buffer system.
Phosphate is most abundant (85%) in bones and teeth. It functions primarily as an intracellular anion and is part of the nucleic acids - DNA, RNA, and ATP-as well as phospholipids and the phosphate buffer system.
Sulfate is part of some amino acids and proteins in the form of sulfur.
|
|
Thread: "Electrolytes 101"
-
12-27-2005, 01:10 AM #1
"Electrolytes 101"

-
12-27-2005, 01:13 AM #2
More related info....
Dr. Philip Maffetone practiced complementary sports medicine and applied kinesiology for over 20 years. His extensive background in biochemistry, kinesiology and exercise physiology has helped him train many world class and professional athletes. He has a doctorate degree in chiropractic and is certified in acupuncture. Athletes he has worked with include Mark Allen, Mike Pigg, Priscilla Welch and Lorraine Moller. Dr. Maffetone was named Coach of the Year in 1996 by Triathlete Magazine. His books include In Fitness and In Health and Training for Endurance (Barmore Productions). He is currently President of the MAF Group, which publishes the Maffetone Report (www.philsbar.com).Water and Electrolytes
by Dr. Philip Maffetone
Water may be the most common nutritional "deficiency" in the running population. This comes in the form of dehydration, even in its common mild forms. Many runners don’t drink enough, and others who perform very long workouts may have a difficult time maintaining normal hydration despite drinking sufficient quantities.
The need to replace water, and the important electrolytes, is much greater than the need to replace any other nutritional substances.
A young man’s body is typically 60% water, and may contain 90 pounds of water; a young woman’s body may contain slightly less at 50% of her total weight. Approximately, two-thirds of this water is in the intracellular compartment of the body — predominantly within the muscles.
The feeling of thirst is activated 1.) when the total body water level is reduced, and 2.) by sodium. Even slight dehydration reduces the blood volume triggering thirst. But thirst is sensed only after dehydration is evident, making thirst a poor indicator that it’s time to drink more water. Once you are dehydrated, it may take up to 48 hours to properly rehydrate using thirst as a guide.
Even mild dehydration reduces blood volume, which reduces blood flow to the muscles, organs and glands, along with oxygen and nutrients since these are also carried in the blood. The ability to expel heat is also lost since skin circulation is reduced, elevating the core temperature of the body. It is vital that runners understand the important functions of proper hydration to avoid its risks, which are amplified in warm weather.
Approximately 60% of the body’s need for water comes from liquids, and 30% from foods. The other 10% is produced in the body from the cellular metabolism. Water loss at rest occurs from the kidneys (60%), from skin and lungs (totaling 30%), sweating (5%) and the large intestine (5%). During running, however, sweating increases significantly, accounting for 90% of the water lost (1-to-2 liters per hour in prolonged training or competition).
If the water lost becomes greater than the intake, blood volume diminishes with significant adverse effects. A runner may, for example, reduce his or her pace by 2% for each percent of body weight lost by dehydration. Water losses of 6-to-10% may exist in marathon events, more in longer competitions. This translates to a runner performing a 10K race in 35 minutes under normal hydration, slowing to complete the same distance in almost 38 minutes when 4% dehydrated – a significant loss of performance.
Under certain conditions, such as during hot weather, the potential for heat stroke is higher. Studies across a number of sports show that mean fluid intakes of up to one liter per hour can realistically be achieved. Therefore, consumption of this amount hourly – in small doses – may serve as a guide during long training and racing.
In athletes with high aerobic function, water regulation is more efficient, including maintenance of body temperatures and lower sweat rates. This is another reason to build a good aerobic base.
For any athlete, it is important to continually drink water everyday and not wait until an upcoming event. Ingestion of large amounts of water at one time, however, may inhibit thirst and actually promote water loss. This may result, over time, in a lower net water volume, and dehydration. The best recommendation is to consume small amount of water throughout the day, everyday. This means one or two cups each time. For most runners, a normal water intake may be between three and four liters per day as a minimum. In athletes who cross train with more total workout time, additional water may be required.
Self-assessing the need for water is important and should be done regularly. Studies have demonstrated the traditional method of assessing for hydration status, by observing the color of the urine, is a good general guide for hydration status. A definite yellow color often indicates dehydration, with a clear urine characterizing proper hydration. Most importantly, runners should learn to assess their own urine color on a daily basis and increase water as necessary.
Electrolytes
A discussion on water intake should also include electrolytes. Sodium and chloride are the dominant electrolytes, along with potassium. Electrolyte regulation is controlled by sodium and chloride, which plays a major role in regulating water.
Dietary intake of sodium, and its loss from the body help regulate both the thirst and salt appetite mechanism in the brain. This is also associated with the actions of kidney hormones which help maintain rehydration for some hours after water ingestion. As a result of this, pre-competition drinks or foods should contain moderate levels of sodium.
The adrenal hormone aldosterone is very important for electrolyte regulation – it prevents sodium and chloride loss, and rids the body of excess potassium. Adrenal stress, however, can result in excess sodium and chloride loss, and increased potassium, resulting in a diminished water volume.
A common problem in runners who have reduced aldosterone is diarrhea, which may cause further losses of sodium and water. The so-called "athlete’s diarrhea" may be due to chronic adrenal dysfunction – a condition which may not be evident until the stress of competition. This is especially true in those competing in longer events. The chronic loss of sodium during adrenal dysfunction has also long been considered the reason for salt craving by many athletes.
Hyponatremia (blood sodium concentration below 136 mmol/L) can occur during or after training or racing, and can sometimes be observed even at rest in a normal blood test. Runners with low sodium do not necessarily have symptoms of hyponatremia. Early symptoms may include weakness or disorientation. In extreme cases, hyponatremia can result in such problems as rapid neurological deterioration, cardiovascular instability and seizures.
The use of sodium during competition may also be important, especially in long endurance events, as hyponatremia can occur if too much water and too little sodium is consumed. Small amounts of sodium added to water (just enough to get a very mild salt taste) can speed stomach emptying and water absorption.
I have recommended sodium tablets to many athletes for use during long endurance events. The well-hydrated runner sucks on a salt tablet until he or she loses the desire/taste for salt. In some cases, a runner may consume several salt tablets during events such as a marathon.
While large amounts of salt have been given to healthy individuals with no adverse effects, some people may be sodium sensitive. In this case, the blood pressure may rise or water retention will be evident.
With so much concern about diet and nutrition, the first step should be to assure you remain well hydrated.
by Dr. Philip Maffetone
http://www.rrca.org/publicat/wat.htmLast edited by cxm; 12-27-2005 at 01:21 AM.

-
12-27-2005, 01:17 AM #3
Related info...
By Joseph BergerRestoring Electrolytes for Athletic Performance
By Joseph Berger
What vitamins have been found to offer benefits for restoring electrolytes and athletic performance?
There are a variety of nutrients shown in studies to provide benefits for restoring energy and improving athletic performance. Of course, taking a good multivitamin – one formulated with optimal amounts and forms of vitamins, minerals, antioxidants, herbs and amino acids – can help to ensure your overall health, from head to toe.
Multivitamins can provide benefits for countless health concerns, including immune, cardiovascular, vision, muscle, bone, and nerve health. And in regards to energy and endurance, these supplements have been shown to provide effective support. But for individual nutrients, the following vitamins have been shown to provide valuable sources of energy and endurance benefits:
• Vitamin C (ascorbic acid) – considered essential for general health and well-being, this multi-functional antioxidant may hold properties for energy and endurance. Deficiencies of this nutrient may be linked to feelings of malaise and generalized fatigue. Several published studies show that after several days of taking vitamin C (400-3,000 mg) daily before and after intense exercise, pain was reduced and muscle strength recovery was rapidly increased. It is thought that as antioxidants may neutralize free radicals before they can cause bodily damage, supplementation of these nutrients may aid in exercise recovery.
• Vitamin E (tocopherol, tocotrienol) – this thoroughly researched antioxidant has been found to slow cellular aging. Additionally, various published studies suggest that daily vitamin E supplementation (800-1,200 IU) may reduce free radical activity and muscle damage caused by strenuous exercise.
• Vitamin B1 (thiamine) – considered vital for every cell, this water-soluble (dissolves in water) B vitamin is essential for producing adenosine triphosphate (ATP), the body’s fuel source. It processes carbohydrates, proteins, and fats, and is crucial for generating metabolic energy. In addition, thiamine is needed for the functioning of the muscles, heart and nervous system. It also promotes growth and good muscle tone.
• Vitamin B2 (riboflavin) – this multi-functional nutrient displays antioxidant properties, and helps convert carbohydrates into the fuel ATP. In addition, riboflavin plays a vital role in metabolizing carbohydrates, fats, amino acids and protein, and also activates folic acid and vitamin B6.
• Vitamin B3 (niacin) – besides playing a vital part in the production of ATP, this essential nutrient helps in more than 200 bodily processes. These include the release of energy from fats, the processing of alcohol, and the formation of hormones and blood cells. Vitamin B3 is crucial to energy metabolism and nervous system health, as well as for regulating cholesterol.
• Vitamin B5 (pantothenic acid) – a necessary factor in cellular energy production, this nutrient also contributes to the production of ATP, the body’s energy source. Pantothenic acid is required for the production of lipids (fats), steroids and neurotransmitters. It is also essential for growth, reproduction, and normal physiological functions, such as the activation of the adrenal glands, which help to combat stress.
• Vitamin B12 (cobalamin) – shown be vital for preventing anemia, this nutrient is crucial for energy production, metabolizing carbohydrates, fat and proteins, and for promoting healthy growth and nervous system function. Plus, it may increase energy and promote growth in children.
• Biotin (vitamin H) – this essential B vitamin helps utilize protein, folic acid, and vitamins B5 and B12, while also metabolizing fat and carbohydrates. And, it has been shown to play a part in the production of ATP, the body’s fuel for energy
Article Source: http://EzineArticles.com/?expert=Joseph_BergerLast edited by cxm; 12-27-2005 at 01:22 AM.

-
12-27-2005, 01:37 AM #4
-
-
12-27-2005, 05:51 AM #5
-
12-31-2005, 07:14 AM #6
-
01-06-2006, 01:44 AM #7
Good read:
http://www.bodybuilding.com/fun/drjoe3.htmFree agent
Research and Development Consultant
11+ Years Experience
-
09-01-2006, 05:49 AM #8
-
-
09-01-2006, 06:54 AM #9
-
09-01-2006, 08:11 AM #10
-
09-01-2006, 08:45 AM #11
-
09-01-2006, 09:10 AM #12
-
-
09-01-2006, 04:05 PM #13
-
10-09-2006, 12:35 PM #14
-
10-09-2006, 12:54 PM #15
-
10-09-2006, 12:59 PM #16
-
-
01-07-2007, 01:24 PM #17
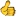
Excellent electrolyte product
http://www.alacer.com/cgi-bin/dbsear...eturntitle.htmEMERGEN-C from Alacer
Nutrition Facts
Serving Size: 1 Packet
Servings Per Container: 36
Amount Per Serving
Calories: 20
Sodium 60mg 3%
Total Carbohydrates 5g 2%
Sugars 5g �
Vitamin C (as Seven Mineral Ascorbates) 1000mg 1111%
Vitamin B1 (as Thiamine HCL) 0.38mg 25%
Vitamin B2 (as Riboflavin) 0.43 25%
Vitamin B3 as Special Niacin Complexes) 5mg 25%
Vitamin B6 (as Pyndoxine HCL) 10mg 500%
Folic Acid 12.5mcg 3%
Vitamin B12 (as Cyanocabalamin) 24mcg 416%
Pantothenic Acid (as Calcium Pantothenate) 2.5mg 25%
Potassium 200mg 6%
Magnesium 60mg 15%
Calcium 50mg 5%
Zinc 2mg 13%
Chromium 10 mcg 8%
Manganese 1mg 8%
Alpha-Lipoic Acid 1mg
-
01-07-2007, 05:19 PM #18
-
01-07-2007, 05:27 PM #19
-
01-07-2007, 05:32 PM #20
-
-
01-07-2007, 05:51 PM #21
-
01-07-2007, 07:55 PM #22ThemoLife Alter Ego

- Join Date: Jul 2002
- Location: Yay Yay Yay Yay Area
- Age: 48
- Posts: 8,160
- Rep Power: 3243












Actually I believe it's the Gatorade Performance Series Endurance Formula
Here's a nice electrolyte product at a nice price.
Hammer Nutrition Endurolytes Powder
cxm needs to include this in his mrp.
Last edited by Supa Freek 420; 01-07-2007 at 08:09 PM.

-
01-07-2007, 09:52 PM #23
-
01-19-2007, 11:15 PM #24
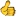
Another impressive electrolyte product here at the BB store, not bad for 9.99 for 32 packs
http://www.bodybuilding.com/store/trace/stamina.html
Trace Minerals Electrolyte Stamina Power Pak
Serving Size: 1 Packet
Servings Per Container: 32
Amount Per Serving:
Calories: 10
Total Fat: 0
Sodium: 134mg
Total Carbohydrates: 2.7mg
Sugars: 2.5mg
Protein: 0
Vitamin C: 1200mg
(as Ascorbic Acid)
Vitamin B2: 1mg
(Riboflavin)
Vitamin B1: 0.45mg
(Thiamin HCL)
Vitamin B6: 10mg
(as Pyridoxine HCL)
Niacin: 5mg
Pantothenic Acid: 3mg
Folic Acid: 20mcg
Calcium: 60mg
(as Calcium Citrate)
Magnesium: 106mg
(as Magnesium Oxide and Concentrace)
Zinc: 3mg
(as Zinc Oxide)
Selenium: 35mcg
(as Selenium Selenate)
Manganese: 1mg
(as Manganese Citrate)
Chromium: 10mcg
(as Chromium Chelavite)
Chloride: 18mg
(from Concentrace)
Potassium: 201mg
(as Potassium Citrate and Concentrace)
Boron: 10mcg
(as Boron Amino Acid Chelate)
Alpha Lipoic Acid: 1 mg
Concentrace: 100mg
(special electrolyte trace mineral blend)
Other Ingredients:
Crystalline Fructose, Stevia, Citric Acid, Lemon Lime Flavor, Sodium Bicarbonate. Plus over 72 naturally occuring trace minerals and elements from Concentrace.
-
-
01-19-2007, 11:19 PM #25
-
01-19-2007, 11:22 PM #26
-
01-20-2007, 12:19 AM #27
-
01-27-2007, 06:31 PM #28Nimbus Nutrition

- Join Date: Feb 2004
- Location: Florida, United States
- Age: 48
- Posts: 708
- Rep Power: 288












 POSEIDON has been released!
POSEIDON has been released!
Poseidon has been released!
http://anabolicminds.com/forum/nimbu...idon-info.html
I'd post more but I don't want to disobey any board rules ;-)
WorkinLIVE ON THE EDGE
-
-
01-27-2007, 06:46 PM #29
You have a very nice physique, but did you have to take this pic wearing no underwear so we could see your glans penis? I mean really, besides the phallis, you look like Justin Bateman on steroids..
http://casadelogo.typepad.com/photos...ed/bateman.jpg
-
01-27-2007, 07:17 PM #30





















Bookmarks